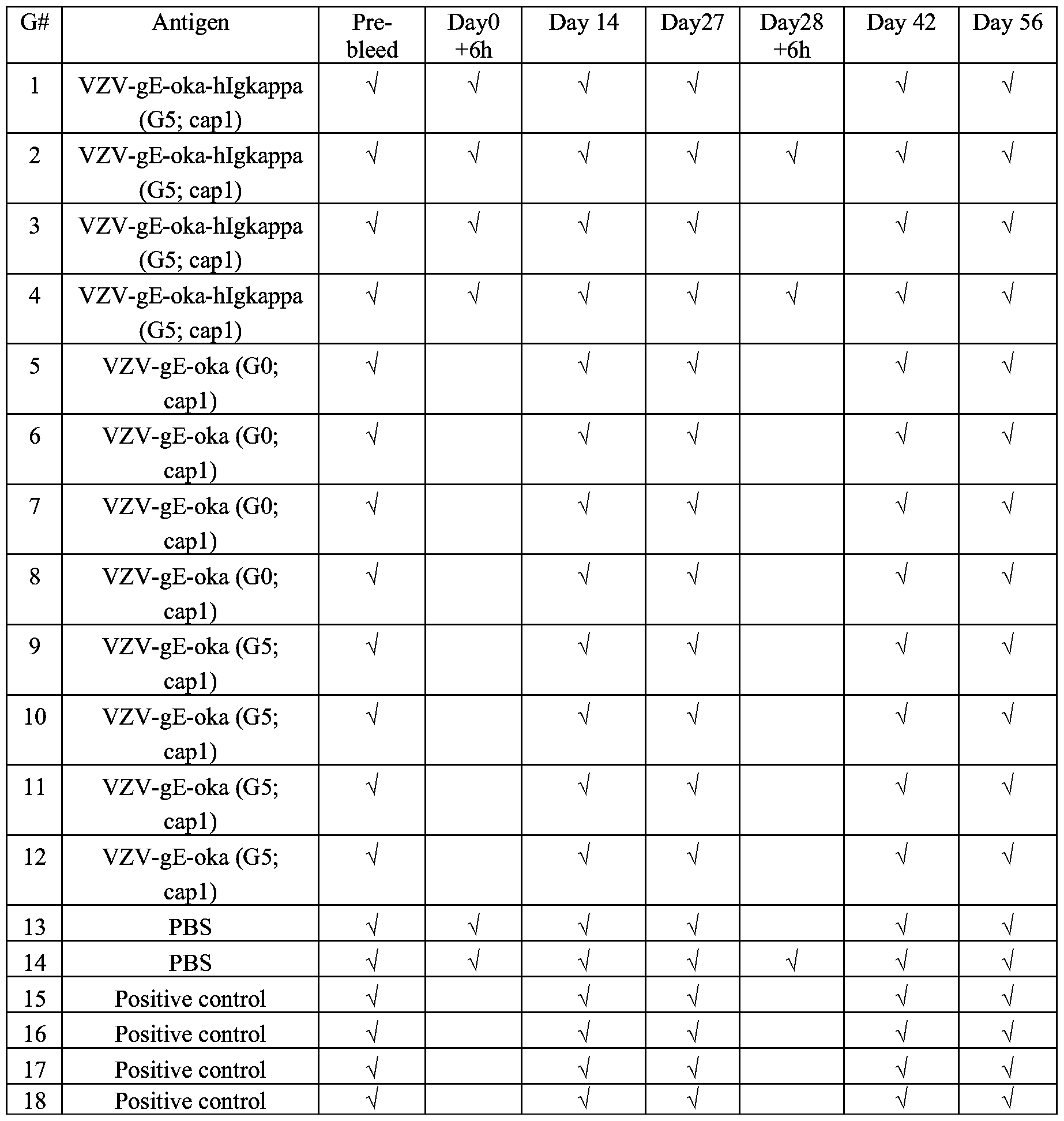
Coverage And Cost Comparison Of Shingrix Vs Zostavax
For adults aged 50 years and older, only plans with Medicare Part D coverage will cover the Shingrix vaccine. However, there may still be a copay even with Medicare Part D coverage. The average cash price for one Shingrix dose is $167, though you may be able to use a prescription discount card to lower this cost. Check with your local pharmacy to see if you can use a Shingrix SingleCare card.
Like Shingrix, Zostavax is primarily covered by Medicare Part D plans or Medicare Advantage plans with Medicare Part D coverage. The copay for Zostavax with insurance can vary. With an average cash price of $278, Zostavax can be expensive with or without insurance. Using a prescription discount card for Zostavax may be able to reduce this cost.
| * |
*not reportedFrequency is not based on data from a head-to-head trial. This may not be a complete list of adverse effects that can occur. Please refer to your doctor or healthcare provider to learn more.Source: DailyMed , DailyMed
Is Shingrix Or Zostavax More Effective
Shingrix and Zostavax have both been proven to prevent shingles. However, Shingrix is a newer vaccine that is considered more effective than Zostavax. Shingrix is even recommended for those who have already received the Zostavax vaccine in the past.
Clinical trials have shown that Shingrix is 97% effective at preventing shingles in adults aged 50 to 69 years old. Shingrix is also effective in preventing shingles in older adultsadults over the age of 70, Shingrix is 91% effective.
Zostavax has a 70% efficacy rate in preventing shingles in adults aged 50 to 69 years old, according to the Zoster Efficacy and Safety Trial . Results from the Shingles Prevention Study showed that Zostavax is 51% effective against shingles. Compared to Shingrix, the effectiveness of Zostavax decreases in older age groups. Based on the SPS results, Zostavax is 64% effective in adults aged 60 to 69 years old 41% effective in adults aged 70 to 79 years old and 18% effective in adults aged 80 years and older.
Your healthcare provider will most likely recommend Shingrix over Zostavax. Shingrix is especially recommended for immunocompromised patients since it is a non-live vaccine. Ask your doctor or pharmacist about which shingles vaccine is right for you.
Pfizer And Biontech To Make Mrna
Pfizer PFE and its partner for COVID-19 vaccine BioNTech BNTX signed a new collaboration to develop an mRNA-based vaccine for the prevention of shingles, a painful and disfiguring disease.
Clinical studies on the mRNA-based shingles vaccine are expected to begin in the second half of 2022. The companies will make the vaccine by leveraging Pfizers antigen technology and BioNTech’s proprietary mRNA platform technology, which has been used to make their successful COVID-19 vaccine.
For the deal, Pfizer will make an upfront payment of $225 million to BioNTech that will include an upfront cash payment of $75 million and an equity investment of $150 million. In addition, BioNTech will also be entitled to receive potential milestone payments of up to $200 million while being eligible for a future share of gross profits, if some products are successfully developed.
This is Pfizer and BioNTechs third mRNA vaccine collaboration. Pfizer and BioNTech have successfully developed an mRNA-based vaccine for COVID-19, under a collaboration initiated in 2020, that is now authorized in several countries and is probably the most successful of all vaccines available. The COVID-19 vaccine has generated billions in revenues for companies. Pfizer and BioNTech also have a collaboration to make an mRNA-based influenza vaccine, which was initiated in 2018.
Pfizers stock has risen 56% in the past year compared with an increase of 22.1% for the industry.
Read Also: Side Effects For Shingles Shot
Design And Expression Of The Mrna Vaccine Encoding Rabv
RABV-G is the major viral antigen that induces neutralizing antibody production in infected species . To both improve its expression and stabilize its desired conformation, we designed a panel of RABV-G mRNA antigens: RABV-G A, RABV-G B, and RABV-G C . We then compared the antigen expression induced by these optimized mRNA constructs. Flow cytometry analysis revealed that the expression of RABV-G induced by RABV-G C was the highest among the three mRNA constructs , suggesting that both sequence optimization and poly tailing are necessary for cellular G protein expression. As lipid shells are necessary for mRNA vaccine delivery , the RABV-G C sequence was then encapsulated into lipid nanoparticles to become our mRNA vaccine candidate LVRNA001. Briefly, the LVRNA001 construct includes a 5 cap structure, a 5 UTR, an open reading frame , a 3 UTR and a poly tail. The ORF in this study encodes the glycoprotein of the CTN-1 strain . Spherical nanoparticles with a uniform size distribution were observed under an TEM .
Fig. 1
Design and expression of the mRNA vaccine encoding RABV-G. A A panel of RABV-G mRNA antigens was designed. B HEK293 cells were transfected with RABV-G mRNA or Luciferase mRNA for 24 h. Cells were collected and used to measure RABV-G expression by flow cytometry. C Spherical nanoparticles were observed under an electron microscope
Pfizer And Biontech Enter Deal To Develop First Mrna Vaccine For Shingles
Pfizer will pay BioNTech $225m in an upfront payment, which includes a cash payment of $75m and an equity investment of $150m.
Pfizer and BioNTech have entered a new research, development and marketing partnership for a potential first mRNA-based vaccine to prevent shingles ).
The partnership builds on the success of the approved mRNA Covid-19 vaccine developed by the companies.
In 2018, Pfizer and BioNTech partnered to develop an influenza vaccine and commenced a collaboration for the Covid-19 vaccine in 2020.
The latest partnership is the third by the companies in the field of infectious diseases.
The parties will leverage Pfizers antigen technology and BioNTechs mRNA platform technology used in their Covid-19 shot.
According to the latest deal, BioNTech is entitled to an upfront payment of $225m from Pfizer.
This comprises a cash payment of $75m and an equity investment worth $150m.
Pfizer will also pay regulatory and sales-based milestone payments worth up to $200m to BioNTech in the future.
Furthermore, Pfizer is eligible for $25m from BioNTech for leveraging Pfizer-identified antigen sequences.
The companies will split the development expenses while the clinical trials are anticipated to commence in the second half of next year.
Pfizer will own the rights to market the potential vaccine across the globe, excluding Germany, Turkey and some developing nations where BioNTech will own the marketing rights.
Also Check: What Does Shingles Look Like On Black Skin
What Will The Clinical Trials For Mrna Flu Vaccines Aim To Show
The trial aims to show whether the new vaccine is safe and whether its immunogenic, which means, Does the vaccine stimulate antibodies that are likely to be protective against influenza?
Researchers will also be studying what the best dose is and whether a single dose is adequate or whether recipients will need two doses.
Why The Shingles Vaccine Can Also Decrease Your Risk Of A Stroke
If you’re like most Americans, you have a distinct memory of being sick at home with chickenpox, per the Mayo Clinic. There was the fever and headaches, oatmeal baths and calamine lotion for itchy skin, and trying to stomach chicken soup even though you didn’t have an appetite. While the virus lasts for about a week, the effects of shingles may last a lifetime.
According to the CDC, about one in three adults will develop shingles. With 95% of American adults having contracted chickenpox in their life, the risk of getting shingles is extremely common. Once chickenpox leaves the body, the virus that causes it, herpes zoster, remains. It can later be reactivated, resulting in shingles, a painful, itchy rash that affects one side of the body.
Although the link between chickenpox and shingles is well-known, the link between shingles and strokes has been less clear. According to the American College of Cardiology, researchers found that shingles increased the risk for stroke by 35%, with the risk being higher for people with shingles who were under 40 years old. A new study now says that taking the shingles vaccine can decrease the risk of a stroke.
Also Check: Where Are Shingle Shots Available
Can The Covid Vaccine Cause Shingles
According to a small study by the University of California External Site, there is currently no evidence that proves the COVID vaccine can cause shingles External Site. However, scientists say that after you get the COVID-19 shot, your immune system gets busy creating antibodies. This means your immune system may become temporarily compromised to other health conditions .
Researchers are still getting a better understanding of how the COVID-19 vaccine impacts a potential shingles outbreak. One clinical trial External Site has even suggested that getting the shingles vaccine may help your body fight off the flu and COVID-19.
Shingles Vaccines For 2022
Shingles is caused by the varicella-zoster virus , the same virus that causes chickenpox, says the U.S. CDC. After a person recovers from chickenpox, the virus stays dormant in the body. However, this virus can reactivate years later, causing shingles. Your risk of shingles increases as you get older, as well as serious complications, says the U.S. NIH.
Immuinize.org published updated FAQs regarding the recombinant, adjuvanted zoster vaccine .
Read Also: Can I Get A Shingles Shot At Walgreens
Related: Gsks New Shingles Vaccine May Win Nod Over Mercks Zostavax
We are absolutely thrilled to get to this point because the science behind this vaccine offers tremendous potential for helping patients, protecting them against getting shingles and its complications, said Dr. Leonard Friedland, vice president for scientific affairs and public health for GSKs North American vaccines unit.
Shingles is a delayed complication of chickenpox, a highly infectious disease most children suffered through in the era before the chickenpox vaccine was developed.
People who have had chickenpox harbor the varicella zoster virus the bug that causes the disease in their bodies after infection.
Healthy immune systems keep the virus in a dormant state. But people who have weakened immune systems, either because of an illness or because the immune system declines with age, can develop shingles.
The condition typically manifests as a painful, itchy rash, either on one side of the torso or one side of the face. The blisters can lead to ulcers and scars.
Theres nothing good about shingles, said Dr. Anne Louise Oaklander, a shingles expert who is director of the nerve unit at Massachusetts General Hospital.
And it can be quite bad. In the worst cases, people may not be able to put their clothing on. And it can leave ulcers and scars.
Shingles can also lead to strokes, vision and hearing loss, and other complications, Oaklander said.
Pfizer And Biontech To Develop Mrna Shingles Vaccine
Posted: 6 January 2022 | Hannah Balfour |
New collaboration between Pfizer and BioNTech will develop an mRNA vaccine to combat shingles .
Pfizer and BioNTech have announced a new research, development and commercialisation collaboration to develop an messenger RNA -based vaccine to prevent shingles, also known as herpes zoster virus .
Building on the success of their COVID-19 vaccine development collaboration, under the new agreement, the companies will leverage a proprietary antigen technology identified by Pfizers scientists and BioNTechs proprietary mRNA platform technology used in the COVID-19 vaccine. The parties will share development costs and expect clinical trials to start in the second half of 2022. Pfizer will have rights to commercialise the potential vaccine on a global basis, with the exception of Germany, Turkey and certain developing countries where BioNTech will have commercialisation rights. The companies will share gross profits from commercialisation of any product.
Under the terms of the agreement, Pfizer will pay BioNTech $225 million in upfront payments, including a cash payment of $75 million and an equity investment of $150 million. BioNTech is eligible to receive future regulatory and sales milestone payments of up to $200 million. BioNTech will pay Pfizer $25 million for the companys proprietary antigen technology.
Recommended Reading: How Do You Know If Its Shingles
Shingrix Use For Immunocompromised Adults
The CDC stated on Feb. 17, 2022: that the Shingrix vaccine is recommended for persons 19 years old and older who have altered immunocompetence. And vaccination of Contacts of Persons with Altered Immunocompetence Household contacts and other close contacts of persons with altered immunocompetence should receive all age- and exposure-appropriate vaccines, except for the smallpox vaccine.
During the U.S. CDC’s Advisory Committee on Immunization Practices meeting on October 20, 2021, Tara Anderson, DVM, MPH, Ph.D., presented: Interpretation of the EtR Regarding Use of RZV in Immunocompromised Adults, Considerations for Use, and Proposed Policy Options. The ACIP’s Recommendation is: Two doses of recombinant zoster vaccine are recommended for adults aged 19 years who are immunodeficient or immunosuppressed due to disease or therapy to prevent herpes zoster and its complications.
Previously, Camille Nelson Kotton, M.D., Chair, ACIP Herpes Zoster Work Group, presented the Introduction Zoster Vaccines Session on September 29, 2021 Ismael R. Ortega-Sanchez, Ph.D. presented ‘Economics of vaccinating immunocompromised 1949-years-old adults against herpes zoster in the USA Tara Anderson, DVM, MPH, Ph.D. presented ‘Preliminary Evidence to Recommendations Framework Regarding Use of Recombinant Zoster Vaccine in Immunocompromised Adults and Next Steps.’
Why You Probably Wont Need A Shingles Booster

Poland and Schaffner both noted that since the effectiveness of Shingrix seems to be lasting, its not likely that people will ever need a shingles booster shot after they receive their primary series, given as two shots six months apart.
At the moment, the recommendation is for everyone aged 50 and older to get this vaccine, said Schaffner. And after youve had your first two dosesso farthere does not appear to be a need for a follow-up.
Poland said that researchers will keep collecting data, as we always do and if there is ever evidence that the vaccine has lost effectiveness, we would know in time to either recommend a booster dosewhich I dont think its going to be likelyor do something else.
Alison Hunt, a spokesperson for GSK, the manufacturer of Shingrix, told Verywell that the company is committed to studying the longer-term efficacy, immunogenicity, and safety of the vaccine which was licensed in 2017.
The Zoster-049 study is still ongoing with final data expected in 2024. Upon study completion, the product information may be updated as appropriate, Hunt said.
Also Check: Does Cvs Offer Shingles Shots
Who Should Get Shingrix
Adults 50 years and older should get two doses of Shingrix, separated by 2 to 6 months. Adults 19 years and older who have or will have weakened immune systems because of disease or therapy should also get two doses of Shingrix. If needed, people with weakened immune systems can get the second dose 1 to 2 months after the first.
You should get Shingrix even if in the past you:
- Received varicella vaccine
There is no maximum age for getting Shingrix.
If you had shingles in the past, Shingrix can help prevent future occurrences of the disease. There is no specific length of time that you need to wait after having shingles before you can receive Shingrix, but generally you should make sure the shingles rash has gone away before getting vaccinated.
Chickenpox and shingles are related because they are caused by the same virus . After a person recovers from chickenpox, the virus stays dormant in the body. It can reactivate years later and cause shingles.
Shingrix is available in doctors offices and pharmacies.
If you have questions about Shingrix, talk with your healthcare provider.
* A shingles vaccine called zoster vaccine live is no longer available for use in the United States, as of November 18, 2020. If you had Zostavax in the past, you should still get Shingrix. Talk to your healthcare provider to determine the best time to get Shingrix.
Great News For Us Bad News For Varicella Zoster
William Schaffner, MD, a professor in the Division of Infectious Diseases at Vanderbilt University Medical Center, told Verywell the findings are great news.
I didnt think it was pretty good dataI thought it was great data. I mean, this is a spectacular vaccine, he said.
Greg Poland, MD, the director of the Vaccine Research Group at the Mayo Clinic and the editor-in-chief of the journal Vaccine, agreed, adding the vaccine is great news for us but bad news for the virus.
Once you have chickenpox, you never get rid of it, said Poland. Its always in your body as a chronic infection.
That may not be a major concern for healthy, young people. However, Poland explained that as we get older, our immune systems start to have a harder time keeping a chronic infection in checkwhats called immunosenescence.
Given the decline in immune function with age, there have been concerns that a shingles vaccine for older people would not be successful. However, according to Poland, the fact that shingles shot can still offer protection 10 years after an older person receives it is a testament to the immunogenicity of the vaccine.
Indeed, the data show that the efficacy rate for Shingrix during the first few years after vaccination is well above 90% and is still between 80% and 90% 10 years later.
Read Also: What Kind Of Pain Do You Get With Shingles
Conditions Treated By Shingrix And Zostavax
Shingrix and Zostavax are FDA approved to prevent shingles . Both vaccines are indicated to prevent shingles in adults aged 50 years and older. Shingrix and Zostavax are not used to prevent primary varicella infection, also known as chickenpox.
Postherpetic neuralgia is a common type of nerve pain that arises with shingles. Because Shingrix and Zostavax can prevent shingles, they can also prevent postherpetic neuralgia and other painful complications from shingles. However, these vaccines are not labeled to treat PHN.
| Condition |
| Yes |